Autologous Cell Therapy
Eliminate cross-contamination risk. Process patient-derived cells in a fully closed, automated system — from gene editing through formulation — with the UniFuge® platform.
The challenge
Multi-product suites managing patient-specific batches need absolute product integrity between runs.
Traditional equipment requires extensive cleaning validation between patient batches, consuming critical processing time.
Open-system interventions introduce contamination risk and operator variability in GMP environments.
Inconsistent processing conditions across patient batches compromise regulatory confidence and product quality.
How it works
Gentle separation concentrates cells and exchanges media, preserving cell integrity before gene editing.
Media exchange and optional washing remove residual media while maintaining cell health for continued growth.
Low-shear separation gently concentrates cells, minimizing damage with optional washing to enhance purity.
Automated washing strategies remove process impurities while maintaining viability above 95%.
Cells are concentrated and media exchanged with formulation buffers, preparing them for cryopreservation with minimal loss.
Full documentation (IQ/OQ, SAT, MVR) and 21 CFR Part 11 compliance support regulatory submissions.
Key benefits




Data highlights
Cell recovery across TIL, NK, iPSC, and stem cells
Cell viability maintained during processing
Viability reduction across all cell types tested
Washing efficiency after just two wash cycles
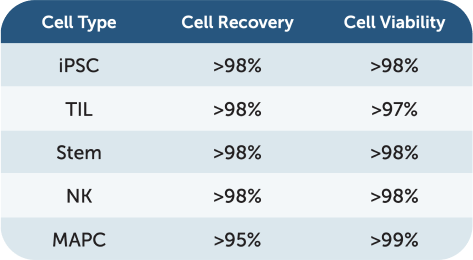
UniFuge delivers over 98% recovery for iPSC, TIL, stem, and NK cells with over 99% viability. For MAPC cells, it ensures over 95% recovery and 99% viability.
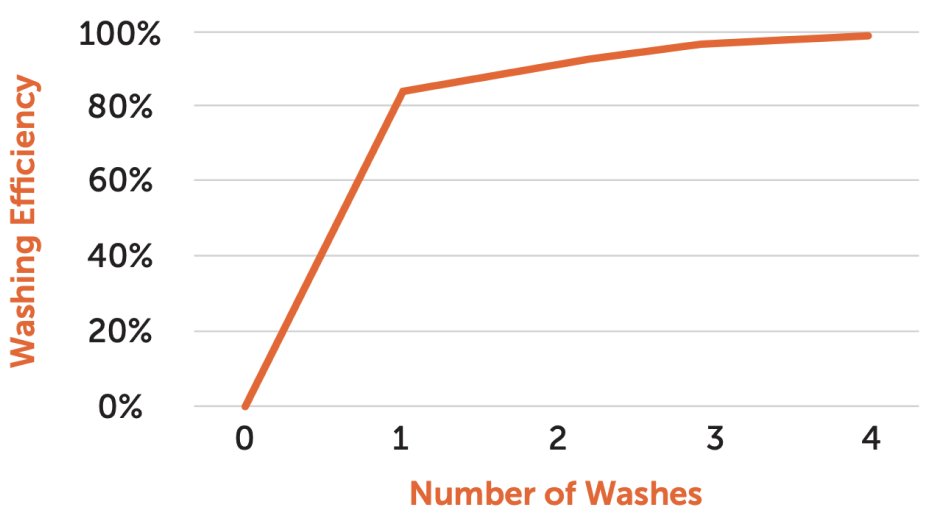
UniFuge demonstrates over 90% washing efficiency after just two washes, effectively removing residual media and additives critical for patient-specific batches.
Case study
During autologous cell therapy manufacturing, maintaining cell viability and recovery during harvesting is critical — but many available technologies fall short. Some provide high recovery but damage cells, while others preserve viability but underperform in recovery.
CARR partnered with a customer to test the UniFuge Pilot and UFMini for cell harvesting across stem cells, NK cells, and MAPCs. Different g-forces (400–1200 xg) and flow rates (100–1,600 mL/min) were evaluated to optimize recovery and minimize viability reduction.
What teams are saying
"This one is perfect. This would reduce our harvest time significantly. I think it fits well into the needs for a Phase III trial."
Cell Therapy Bioproduction Expert
"The UniFuge was fairly simple to use and scaled nicely from the UFMini."
Cell Therapies Bioproduction Expert
"The UniFuge line is simpler and required less calibration than other solutions we have tried."
Protein Separation Bioproduction Expert
Centrifuges

Small-to-mid-scale processing. High cell recovery with seamless scaling to larger production.
Learn more

Early-stage development. Low-shear, high-throughput processing with up to 98% cell viability.
Learn more

Large-scale commercial production. High-throughput clarification and harvesting at over 2,000 L.
Learn more
Stay connected
Connect with a CARR expert to discuss how the UniFuge platform can streamline your patient-specific manufacturing — from process development through commercial scale.